(厦门大学材料学院,福建省高校生物医学工程重点实验室,福建 厦门 361005)
(Key Laboratory of Biomedical Engineering of Fujian Province,College of Materials,Xiamen University,Xiamen 361005,China)
DOI: 10.6043/j.issn.0438-0479.202009002
备注
引言
20世纪60年代中期,病理学家在被病毒感染的人类和动物组织样本中首次观察到类病毒纳米颗粒[1-3],这些纳米颗粒与病毒形态相似但没有传染性,将其称为病毒样颗粒(virus-like particles,VLPs).进一步研究发现,VLPs是一种由病毒蛋白重复排列而成的规则的中空蛋白颗粒结构,能够展示病毒表位,但不包含病毒遗传物质,不具有复制和感染特性,多数呈球状或管状[4].相比于其他纳米载体,VLPs具有以下几点优势:1)通过重组蛋白表达即可实现简单、经济、可控的大规模生产; 2)组成和结构均一,易于在特定位置进行功能化改造; 3)在体内外均具有生物相容性和生物可降解性; 4)解聚重组能力和空腔结构易于进行药物包载[5-6].这些特性引起了人们对开发VLPs作为疫苗和药物递送载体的浓厚兴趣.大量研究证实VLPs具有较高的免疫原性,能够充分激活机体免疫反应,促进B细胞活化并释放抗体[4,7-13],并且纳米尺度的VLPs可以被抗原递呈细胞(antigen-presenting cells,APCs)大量摄取并降解,从而活化T细胞[14-16].VLPs除了作为疫苗应用外,利用VLPs的空腔结构和可修饰性可以包裹或展示一些小分子药物,从而实现靶向递送,极大改善了药物的体内分布和疗效[6,17-20].在过去的50多年里,VLPs已经成为生物医学研究中的重要工具(图1)[21-26].本文综述了VLPs在肿瘤治疗中的研究进展,着重介绍了VLPs作为肿瘤药物递送载体和疫苗的设计.
1 VLPs的免疫原性
T细胞在抗癌免疫反应中发挥着重要作用.APCs通过免疫组织相容性复合物(major histocompatibility complex,MHC)可以将抗原递呈给T细胞识别,其中由MHC Ⅰ递呈的抗原可以刺激并活化CD8+细胞毒性T细胞(cytotoxic T lymphocytes,CTL)直接
杀伤肿瘤细胞,此外,CTL产生的细胞因子能够形成促炎症环境进而杀伤肿瘤细胞[27].由MHC Ⅱ递呈的抗原可以刺激并活化CD4+T细胞,该细胞的不同亚型在抗肿瘤反应中的作用不同:Th1(CD3+CD4+干扰素γ(IFNγ)+)细胞通过分泌促炎症细胞因子,辅助CD8+CTL裂解肿瘤细胞[28]; 而另一种免疫抑制细胞Tregs(CD3+CD4+Foxp3+)则通过分泌白介素10(IL-10)和肿瘤生长因子β(TGF-β)等细胞因子,抑制CTL增殖并降低炎症反应[29-30].因此,加强Th1细胞和CTL的激活是抗肿瘤疫苗研制的关键.VLPs能够引起一定固有免疫,且易于进行基因工程改造[31-36],已被证实可以有效诱导Th1细胞和CTL的增殖和分化[37-39],因此通过基因工程改造VLPs或用VLPs包裹免疫刺激类药物即可实现多种免疫反应协同抗肿瘤的疗效.已有研究表明,展示T细胞表位的VLPs或将VLPs与APC刺激剂联合使用可刺激有效的Th1细胞和CTL应答.例如:当噬菌体衍生的Qβ-VLP携带Toll样受体(Toll-like receptor,TLR)的配体(如RNA或富含未甲基化CG基序的寡核苷酸(oligonucleotides rich in unmethylated CG motifs,CpGs)等)时,它们可以刺激CTL和Th1细胞应答; 但当它们携带多聚谷氨酸(polyGLU)时,则无法检测到有效的应答[40-41].由于VLPs的特性,一些内核富含精氨酸的VLPs(如HBcAg VLPs)在宿主细胞中表达时会聚集在核酸周围[41],这也为VLPs作为核酸递送载体奠定了一定基础.目前,VLPs可以用于递送不同的TLR配体,如CpGs(TLR-9配体)、polyGLU、单链核糖核酸(single-stranded ribonucleic acid,ssRNA)(TLR 7/8配体)或双链核糖核酸(double-stranded ribonucleic acid,dsRNA)(TLR-3配体),并且VLPs可以在这些TLR配体存在的情况下实现解聚和重组,解聚时大量配体被正电性的精氨酸富集末端吸引,重组后包裹进VLPs内部[42-46].
VLPs除了能够引起T细胞相关的反应外,也能够引起B细胞相关的免疫反应.VLPs被认为是一种功能强大的病原相关分子模式(pathogen-associated molecular patterns,PAMP),它能被B细胞的受体识别,并使B细胞足以独立激活CD4+T细胞并长时间维持其功能[7,47].夏宁邵教授团队[48-49]应用大肠杆菌(Escherichia coli)原核表达系统研制出多种VLPs疫苗,如2012年上市的全球首个戊肝疫苗和2020年上市的人乳头瘤病毒(HPV)疫苗(即宫颈癌疫苗),在真核表达系统之外开辟了疫苗研究的新途径.VLPs高度重复的结构可以被多个B细胞受体(B-cell receptor,BCR)识别并向B细胞传递刺激信号,在引起B细胞免疫反应中有很大优势[50-51].此外,VLPs表面可用于展示多达300~400个肽段,诱导抗体反应[52].VLPs还能展示肿瘤细胞表面的特异性抗原人表皮生长因子受体-2(human epidermal growth factor receptor-2,HER2)蛋白,以诱导B细胞反应,刺激特异性抗体的产生,最终提高肿瘤组织的炎症水平,增强CD4+和CD8+T细胞的活性[53-54].
VLPs类似于天然病毒,通常比亚单位或重组蛋白免疫原更具免疫原性[55-56].研究发现VLPs可以被巨噬细胞和树突状细胞(dendritic cells,DCs)等细胞摄取,激活固有免疫,之后大量细胞因子的释放和抗原的交叉递呈可以进一步激活适应性免疫反应[37].VLPs的球状粗糙多凸起的表面,兼具流动性和柔韧性,利于抗原受体结合,是被APCs有效摄取的关键[57].VLPs被APCs摄取加工后可以被MHC Ⅱ或MHCⅠ分子呈递于细胞表面,同时还能刺激DCs成熟并迁移到淋巴结递呈抗原,从而激活T细胞.此外,外来抗原一般只能由MHC Ⅱ分子递呈[58-59],而VLPs与其他外源抗原不同,它可在APCs内经历交叉提呈,在没有感染细胞的情况下也能有效地被MHC Ⅰ分子递呈[60-61].因此,VLPs能有效刺激CD4+ T淋巴细胞和CTL增殖.综上,VLPs作为肿瘤疫苗具有先天优势,其免疫原性可以有效激活机体产生免疫反应,但是作为药物递送载体时往往需要通过一些手段去降低VLPs的免疫原性,使其在体内能有效递送药物.
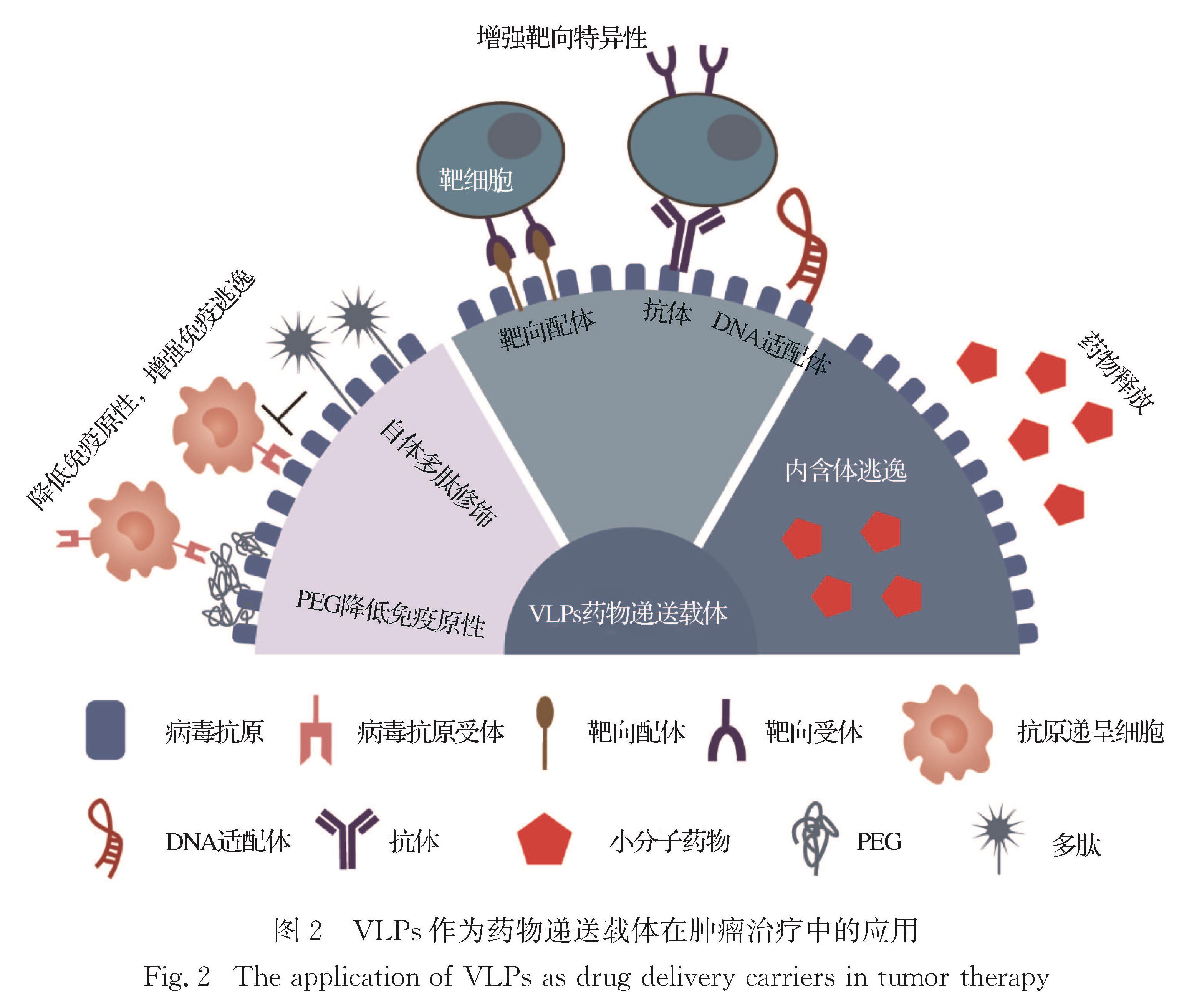
2 VLPs作为药物递送载体在肿瘤治疗中的应用
VLPs的粒径有利于细胞胞吞,还兼具生物可降解性和安全性,通过基因工程或化学修饰即可对其3个不同界面(蛋白颗粒的外部、内部和亚基之间)进行功能化改造[39],这使其成为理想的药物递送载体(图 2).Ao等[62]利用人类免疫缺陷病毒群体特异性抗原(HIV-Gag)可自组装成VLPs这一特点,设计了融合半胱氨酸蛋白酶8(caspase 8)的Gag-CASP8融合蛋白,并与水泡性口炎病毒G糖蛋白(VSV-G)形成伪病毒Gag-CASP8-VLPs.VSV-G可介导颗粒有效进入细胞,增强其在胞浆内传递,有效地将caspase 8递送到细胞内部,诱导肿瘤细胞凋亡,进而抑制乳腺肿瘤的生长.
病毒结构蛋白内部常带有对核酸具有亲和性的氨基酸残基,以实现更稳定地包裹病毒遗传物质的作用.因此,以病毒结构蛋白为主体的VLPs可作为基因药物的良好载体[63].Lin等[64]使用人JC多瘤病毒(JCPyV)来源的VLPs作为基因递送载体,将携带肺癌特异性自杀基因启动子(SP-B)和CD59特异性短发夹RNA(shRNA)的表达质粒(pSPB-shCD59)有效地递送到肺癌细胞内,增强了补体依赖性细胞毒性,并有效地抑制了肺部肿瘤的生长.本课题组已实现了VLPs表面氨基酸序列的靶向改造,如在VLPs表面融合表达脑靶向肽(TGN)[65]或整合素受体靶向肽(RGD)[66],同时通过共价或非共价作用装载治疗分子,从而更有效和精准地将小分子化疗药物递送至肿瘤部位,实现良好的抗肿瘤疗效.
VLPs作为肿瘤影像探针平台具有一定的临床应用前景,多种病毒来源的VLPs诊疗平台也已有多方面深入研究[67-68].如有研究者将多种光敏剂、荧光染料等装载到酸浆斑驳病毒(PhMV)来源的VLPs内部或修饰在VLPs表面,VLPs不仅能有效地提高荧光药物的化学稳定性,还能增加肿瘤细胞对药物的摄取[69].Kines等[70]将酞菁光敏剂(IRDye 700DX)偶联的HPV VLPs应用于肿瘤的治疗,在体外实验中发现功能化VLPs(AU-011)能够有效抑制人源肿瘤细胞的生长,在小鼠和兔黑色素瘤皮下模型中均展示出优异的抗肿瘤疗效.Ji等[71]利用铁蛋白纳米颗粒的组装特性,将6-羧基荧光素(FAM)标记的FAP-α(fibroblast activation protein-α)底物肽及其对应的猝灭剂(BHQ)分别偶联至铁蛋白纳米颗粒,成功制备了体内响应纳米荧光探针,用于FAP-α酶高表达的肿瘤检测.本课题组[72]将光敏剂吲哚菁绿(ICG)包载到表面融合了RGD的乙肝病毒核心蛋白(hepatitis B virus core protein,HBc)VLPs内部,不但可作为荧光和光声成像的影像探针,还利用ICG的光动力和光热作用实现了肿瘤的有效治疗.
在VLPs表面用长链高分子进行修饰可以起到有效延长体内循环时间、降低免疫原性、增加稳定性、增强渗透性和滞留(enhanced permeability and retention,EPR)效应等作用[73].Shukla等[74]使用PEG修饰马铃薯X病毒(PVX)和豇豆花叶病毒(CPMV)的VLPs,以降低其免疫原性,结果表明,修饰后的VLPs在小鼠异位肿瘤部位有更高的渗透和积聚,PEG的修饰增强了VLPs的肿瘤归巢和EPR效应,使其成为有潜力的癌症药物递送和成像应用平台.
综上所述,由于VLPs内部可以包裹和保护敏感化合物,外部可以进行修饰,具有容易被修饰的优点,所以VLPs作为药物载体可以实现精确阵列展示大分子或负载小分子药物.良好生物相容性和易于改造修饰的特性使VLPs发展成为一种良好的靶向药物递送系统,有助于进一步扩展和改进现有的药物递送载体体系.
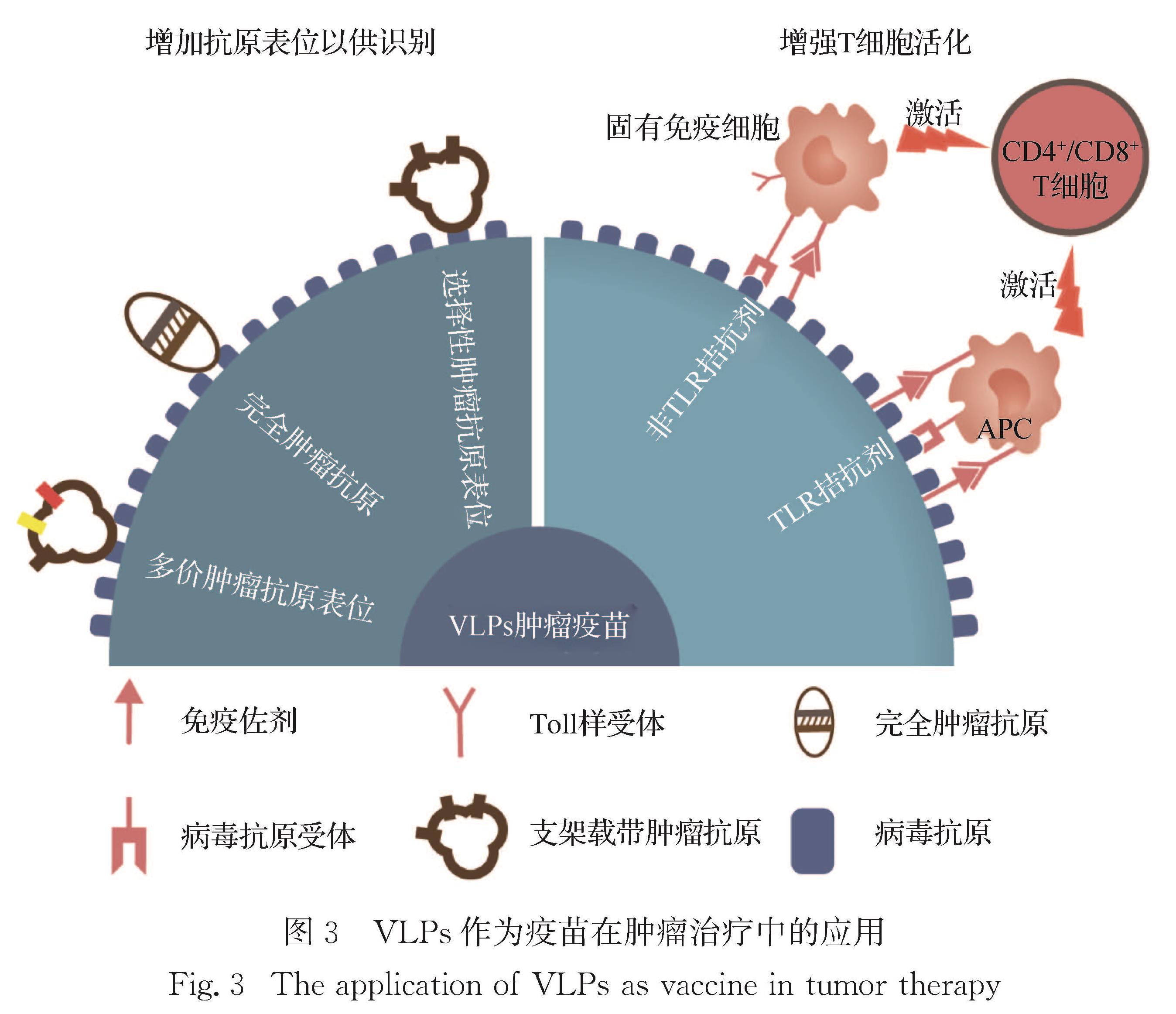
3 VLPs作为疫苗在肿瘤治疗中的应用
VLPs具有天然的免疫原性,以其作为肿瘤疫苗被广泛开发和设计(图 3).在其表面展示外源抗原的技术已被用于生产针对病原体的疫苗,并可有效地打破自身免疫耐受状态,从而启动对功能失调蛋白质的免疫反应[75-76].疫苗接种和其他形式的免疫治疗对于癌症和慢性病的预防和临床治疗上已经有很多应用[75-78].通过基因工程技术,在VLPs多肽结构单元的基因中嵌入一些功能蛋白基因片段,如将VLPs多肽结构单元与抗原融合表达,实现体内的肿瘤免疫治疗.Zheng等[79]通过将CD274的siRNA整合到HPV16-L1蛋白中,构建了一种基于具有免疫检查点阻断功能的纳米疫苗.HPV16-L1蛋白可以激活Ⅰ型IFN介导的先天免疫,siRNA可有效降低免疫检查点CD274的表达,从而保护T细胞的活化和增殖,进而实现协同治疗效果.Hoopes等[80]将来源于CPMV的VLPs(CMV)与放射治疗相结合用于黑色素瘤模型的治疗,在放疗方案中加入CMV不仅可增加肿瘤内免疫细胞浸润,而且还可以显著提高肿瘤局部的炎症环境,进而放大肿瘤释放抗原所产生的免疫效果,显著抑制肿瘤生长,并且能有效抑制肿瘤的转移.
同样地,VLPs的表面还可以与肿瘤抗原进行融合表达,作为肿瘤疫苗进行免疫治疗.Kurg等[81]将3种不同的黑色素瘤抗原MAGEA4、MAGE10和MCAM蛋白融合在逆转录病毒Gag蛋白中,自组装形成VLPs,该重组抗原在哺乳动物细胞中能够被递呈,表明携带重组抗原的逆转录病毒Gag VLPs有望激活肿瘤抗原特异性免疫反应.Simons等[82]用前列腺干细胞抗原(PSCA)和前列腺酸性磷酸酶(PAP-1和PAP-2)这2种前列腺癌相关肿瘤抗原,以及前列腺癌特异性T细胞刺激物(SPAS-1)的多肽修饰牛乳头瘤病毒L1蛋白,与病毒上的半胱氨酸残基形成二硫键,组成新型VLPs疫苗; 该疫苗单独或联合PD1抗体治疗可显著抑制肿瘤生长,提高肿瘤组织中CD3+和CD8+T细胞浸润,表明牛乳头瘤VLPs是一种在转移性前列腺癌患者中具有应用潜力的疫苗载体.Palladini等[54]通过共价修饰,将完整的HER2的胞外结构域与来源于化脓性链球菌(Streptococcus)的纤维连接蛋白SpyCatcher制备成融合抗原,并将融合抗原(SpyCatcher-HER2)附着到AP205噬菌体来源的VLPs表面(表面融合有SpyCatcher对应的标签SpyTag); 该疫苗有效地克服了B细胞的免疫耐受,产生高滴度且具有治疗效力的HER2抗体,有效防止HER2呈阳性的乳腺肿瘤生长,在预防和治疗自发型HER2阳性肿瘤中具有重要意义.该研究结果也证实了利用VLPs平台来开发非传染性疾病疫苗的优越性.
铁蛋白纳米颗粒作为药物递送体系被广泛研究[83].Wang等[84]将分子胶水技术结合到铁蛋白纳米颗粒的设计中,成功制备出能够快速展示病毒或肿瘤抗原的铁蛋白纳米疫苗平台; 该纳米疫苗能够引流至淋巴结并被DCs大量摄取,增强了抗原特异性免疫反应,最终有效抑制HPV16和MC38肿瘤的生长.本课题组[85]将模式抗原鸡卵白蛋白片段(OVA275-264)通过基因工程技术展示在HBc VLPs表面; OVA-HBc VLPs免疫小鼠后,其体内能够产生较强的CTL反应.OVA-HBc VLPs作为肿瘤预防性疫苗,可显著延缓肿瘤生长; 作为治疗性疫苗,在联合低剂量紫杉醇(paclitaxel,PTX)后,展现出有效的抗肿瘤作用.
VLPs疫苗已在人群中广泛接种,并经受了时间的考验,具有生物安全性高、转化潜力大的优点.与减毒疫苗相比,VLPs的另一个优点是可进行多功能化修饰,包括化学结合、基因重组等方法,通过与其他疾病的免疫原性分子结合即可获得具有高免疫原性的疫苗.由于VLPs不通过感染进入宿主细胞,并且抗载体的免疫对VLPs疫苗影响很小[86],使其在肿瘤治疗中具有乐观的应用前景.
4 总结与展望
基于VLPs的可修饰性、稳定性、生产简单和独特的免疫原性,VLPs在肿瘤疫苗开发和药物递送领域具有良好的应用前景.一方面,VLPs的可修饰性和空腔结构可用于抗肿瘤药物的体内递送,对VLPs进行靶向修饰可实现药物的靶向富集; 也可用PEG等免疫惰性物质修饰VLPs来降低免疫原性,从而促进药物在体内的循环时间; VLPs通过一定的工程化改造(如插入酶响应多肽)即可实现药物在靶位置的控制释放.另一方面,VLPs本身就具有较高的免疫原性,可引起机体的先天免疫,偶联或包载免疫刺激物(免疫佐剂)可进一步提高机体的炎症水平,增强T细胞活化; VLPs融合肿瘤抗原后还能引发肿瘤抗原特异性免疫反应,进一步提高抗肿瘤疗效.总而言之,嵌合型VLPs的设计在很大程度上解决了肿瘤抗原和抗癌药物的递送问题.VLPs是一种极其强大的“分子工具箱”,已经在肿瘤治疗中取得可喜的进展,在未来的设计中应该充分发挥VLPs目前已有的优势,融合更多的学科技术丰富其功能,从而加快基于VLPs的肿瘤治疗的智能化、个性化发展.
- [1] BLUMBERG B S,ALTER H J,VISNICH S.A "new" antigen in Leukemia sera[J].JAMA,1965,191(7):541-546.
- [2] MILLMAN I,LOEB L A,BAYER M E,et al.Australia antigen(a hepatitis-associated antigen):purification and physical properties[J].The Journal of Experimental Medicine,1970,131(6):1190-1199.
- [3] BAYER M E,BLUMBERG B S,WERNER B.Particles associated with Australia antigen in the sera of patients with leukaemia,Down's syndrome and hepatitis[J].Nature,1968,218(5146):1057-1059.
- [4] QIAN C Y,LIU X L,XU Q,et al.Recent progress on the versatility of virus-like particles[J].Vaccines,2020,8(1):139.
- [5] MATEU M G.Assembly,engineering and applications of virus-based protein nanoparticles[J].Advances in Experimental Medicine and Biology,2016,940:83-120.
- [6] MA Y J,NOLTE R J M,CORNELISSEN J J L M.Virus-based nanocarriers for drug delivery[J].Advanced Drug Delivery Reviews,2012,64(9):811-825.
- [7] WANG J W,RODEN R B.Virus-like particles for the prevention of human papillomavirus-associated malignan-cies[J].Expert Review of Vaccines,2013,12(2):129-141.
- [8] RYNDA-APPLE A,PATTERSON D P,DOUGLAS T.Virus-like particles as antigenic nanomaterials for inducing protective immune responses in the lung[J].Nanomedicine,2014,9(12):1857-1868.
- [9] PATTERSON D P,RYNDA-APPLE A,HARMSEN A L,et al.Biomimetic antigenic nanoparticles elicit controlled protective immune response to influenza[J].ACS Nano,2013,7(4):3036-3044.
- [10] RICHERT L E,RYNDA-APPLE A,HARMSEN A L,et al.CD11c+cells primed with unrelated antigens facilitate an accelerated immune response to influenza virus in mice[J].European Journal of Immunology,2014,44(2):397-408.
- [11] WILEY J A,RICHERT L E,SWAIN S D,et al.Inducible bronchus-associated lymphoid tissue elicited by a protein cage nanoparticle enhances protection in mice against diverse respiratory viruses[J].PLoS One,2009,4(9):e7142.
- [12] HALPERIN S A,WARD B,COOPER C,et al.Comparison of safety and immunogenicity of two doses of investigational hepatitis B virus surface antigen co-administered with an immunostimulatory phosphorothioate oligodeoxyribo-nucleotide and three doses of alicensed hepatitis B vaccine in healthy adults 18-55 years of age[J].Vaccine,2012,30(15):2556-2563.
- [13] HUBER B,SCHELLENBACHER C,JINDRA C,et al.A chimeric 18L1-45RG1 virus-like particle vaccine cross-protects against oncogenic α-7 human papillomavirus types[J].PLoS One,2015,10(3):e0120152.
- [14] LENZ P,DAY P M,PANG Y Y,et al.Papillomavirus-like particles induce acute activation of dendritic cells[J].Journal of Immunology,2001,166(9):5346-5355.
- [15] LEE S H,CHU K B,KANG H J,et al.Virus-like particles containing multiple antigenic proteins of Toxoplasma gondii induce memory T cell and B cell responses[J].PLoS One,2019,14(8):e0220865.
- [16] STROH E,FISCHER K,SCHWAIGER T,et al.Henipavirus-like particles induce a CD8 T cell response in C57BL/6 mice[J].Veterinary Microbiology,2019,237:108405.
- [17] ZDANOWICZ M,CHROBOCZEK J.Virus-like particles as drug delivery vectors[J].Acta Biochimica Polonica,2016,63(3):469-473.
- [18] REN Y C,MU Y,JIANG L,et al.Multifunctional TK-VLPs nanocarrier for tumor-targeted delivery[J].International Journal of Pharmaceutics,2016,502(1/2):249-257.
- [19] ELZOGHBY A O,SAMY W M,ELGINDY N A.Protein-based nanocarriers as promising drug and gene delivery systems[J].Journal of Controlled Release,2012,161(1):38-49.
- [20] REN Y P,WONG S M,LIM L Y.Folic acid-conjugated protein cages of a plant virus:a novel delivery platform for doxorubicin[J].Bioconjugate Chemistry,2007,18(3):836-843.
- [21] MCALEER W J,BUYNAK E B,MAIGETTER R Z,et al.Human hepatitis B vaccine from recombinant yeast[J].Nature,1984,307(5947):178-180.
- [22] MANCHESTER M,SINGH P.Virus-based nanopar-ticles(VNPs):platform technologies for diagnostic imaging[J].Advanced Drug Delivery Reviews,2006,58(14):1505-1522.
- [23] SCHWARZ B,DOUGLAS T.Development of virus-like particles for diagnostic and prophylactic biomedical applications[J].WIRES Nanomedicine and Nano-biotechnology,2015,7(5):722-735.
- [24] SHEN L H,ZHOU J,WANG Y X,et al.Efficient encap-sulation of Fe3O4 nanoparticles into genetically engineered hepatitis B core virus-like particles through a specific interaction for potential bioapplications[J].Small,2015,11(9/10):1190-1196.
- [25] WNK M,GÓRZNY M L,WARD M B,et al.Fabrication and characterization of gold nano-wires templated on virus-like arrays of tobacco mosaic virus coat proteins[J].Nanotechnology,2013,24(2):025605.
- [26] MAITY B,FUJITA K,UENO T.Use of the confined spaces of apo-ferritin and virus capsids as nanoreactors for catalytic reactions[J].Current Opinion in Chemical Biology,2015,25:88-97.
- [27] HADRUP S,DONIA M,STRATEN P T.Effector CD4 and CD8 T cells and their role in the tumor microenviron-ment[J].Cancer Microenvironment,2013,6(2):123-133.
- [28] KNUTSON K L,DISIS M L.Tumor antigen-specific T helper cells in cancer immunity and immunotherapy[J].Cancer Immunology Immunotherapy,2005,54(8):721-728.
- [29] FACCIABENE A,MOTZ G T,COUKOS G.T-regulatory cells:key players in tumor immune escape and angiogenesis[J].Cancer Research,2012,72(9):2162-2171.
- [30] WANG Y,MA Y S,FANG Y,et al.Regulatory T cell:a protection for tumour cells[J].Journal of Cellular and Molecular Medicine,2012,16(3):425-436.
- [31] DEML L,SCHIRMBECK R,REIMANN J,et al.Recom-binant human immunodeficiency Pr55gagvirus-like particles presenting chimeric envelope glycoproteins induce cytotoxic T-cells and neutralizing antibodies[J].Virology,1997,235(1):26-39.
- [32] KAZAKS A,BALMAKS R,VORONKOVA T,et al.Melanoma vaccine candidates from chimeric hepatitis B core virus-like particles carrying a tumor-associated MAGE-3 epitope[J].Biotechnology Journal,2008,3(11):1429-1436.
- [33] RUEDA P,MARTÍNEZ-TORRECUADRADA J L,SARRASECA J,et al.Engineering parvovirus-like particles for the induction of B-cell,CD4+ and CTL responses[J].Vaccine,1999,18(3/4):325-332.
- [34] TSUNETSUGU-YOKOTA Y,MORIKAWA Y,ISOGAI M,et al.Yeast-derived human immunodeficiency virus type 1 p55gag virus-like particles activate dendritic cells(DCs)and induce perforin expression in Gag-specific CD8+ T cells by cross-presentation of DCs[J].Journal of Virology,2003,77(19):10250-10259.
- [35] BUONAGURO L,TORNESELLO M L,GALLO R C,et al.Th2 polarization in peripheral blood mononuclear cells from human immunodeficiency virus(HIV)-infected subjects,as activated by HIV virus-like particles[J].Journal of Virology,2009,83(1):304-313.
- [36] BUONAGURO L,TORNESELLO M L,TAGLIAMONTE M,et al.Baculovirus-derived human immunodeficiency virus type 1 virus-like particles activate dendritic cells and induce ex vivo T-cell responses[J].Journal of Virology,2006,80(18):9134-9143.
- [37] LIZOTTE P H,WENA M,SHEEN M R,et al.In situ vaccination with cowpea mosaic virus nanoparticles suppresses metastatic cancer[J].Nature Nanotechnology,2016,11(3):295-303.
- [38] BRUNE K D,LENEGHAN D B,BRIAN I J,et al.Plug-and-display:decoration of virus-like particles via isopeptide bonds for modular immunization[J].Scientific Reports,2016,6:19234.
- [39] SAHANDI ZANGABAD P,KARIMI M,MEHDIZADEH F,et al.Nanocaged platforms:modification,drug delivery and nanotoxicity.Opening synthetic cages to release the tiger[J].Nanoscale,2017,9(4):1356-1392.
- [40] KELLER S A,BAUER M,MANOLOVA V,et al.Cutting edge:limited specialization of dendritic cell subsets for MHC class Ⅱ-associated presentation of viral particles[J].The Journal of Immunology,2010,184(1):26-29.
- [41] SCHWARZ K,MEIJERINK E,SPEISER D E,et al.Efficient homologous prime-boost strategies for T cell vaccination based on virus-like particles[J].European Journal of Immunology,2005,35(3):816-821.
- [42] GOMES A C,ROESTI E S,EL-TURABI A,et al.Type of RNA packed in VLPs impacts IgG class switching:implications for an influenza vaccine design[J].Vaccines,2019,7(2):47.
- [43] KAWANO M,MATSUI M,HANDA H.Technologies that generate and modify virus-like particles for medical diagnostic and therapy purposes[M]∥Design and development of new nanocarriers.[S.l.]:Elsevier,2018:555-594.
- [44] DASH M,CHIELLINI F,OTTENBRITE R M,et al.Chitosan:a versatile semi-synthetic polymer in biomedical applications[J].Progress in Polymer Science,2011,36(8):981-1014.
- [45] SIOUD M.Innate sensing of self and non-self RNAs by Toll-like receptors[J].Trends in Molecular Medicine,2006,12(4):167-176.
- [46] STORNI T,BACHMANN M F.Loading of MHC class Ⅰ and Ⅱ presentation pathways by exogenous antigens:a quantitative in vivo comparison[J].Journal of Immunology,2004,172(10):6129-6135.
- [47] MORÓN V G,RUEDA P,SEDLIK C,et al.In vivo,dendritic cells can cross-present virus-like particles using an endosome-to-cytosol pathway[J].Journal of Immuno-logy,2003,171(5):2242-2250.
- [48] XIA L,XIAN Y F,WANG D N,et al.A human monoclonal antibody against HPV16 recognizes an immunodominant and neutralizing epitope partially overlapping with that of H16.V5[J].Scientific Reports,2016,6(1):19042.
- [49] XU L,HE D,YANG L,et al.A broadly cross-protective vaccine presenting the neighboring epitopes within the VP1 GH loop and VP2 EF loop of enterovirus 71[J].Scientific Reports,2015,5(1):12973.
- [50] BACHMANN M F,ZINKERNAGEL R M.Neutralizing antiviral B cell responses[J].Annual Review of Immunology,1997,15(1):235-270.
- [51] BRUN A,BRCENA J,BLANCO E,et al.Current strategies for subunit and genetic viral veterinary vaccine development[J].Virus Research,2011,157(1):1-12.
- [52] PUMPENS P,GRENS E.HBV core particles as a carrier for B cell/T cell epitopes[J].Intervirology,2001,44(2/3):98-114.
- [53] NIKA L S,CUADRADO-CASTANO S,ASTHAGIRI ARUNKUMAR G,et al.An HER2-displaying virus-like particle vaccine protects from challenge with mammary carcinoma cells in a mouse model[J].Vaccines,2019,7(2):41.
- [54] PALLADINI A,THRANE S,JANITZEK C M,et al.Virus-like particle display of HER2 induces potent anti-cancer responses[J].Oncoimmunology,2018,7(3):e1408749.
- [55] HINTON H J,JEGERLEHNER A,BACHMANN M F.Pattern recognition by B cells:the role of antigen repetitiveness versus Toll-like receptors[J].Current Topics in Microbiology and Immunology,2008,319:1-15.
- [56] BACHMANN M F,ROHRER U H,KÜNDIG T M,et al.The influence of antigen organization on B cell res-ponsiveness[J].Science,1993,262(5138):1448-1451.
- [57] BACHMANN M F,JENNINGS G T.Vaccine delivery:a matter of size,geometry,kinetics and molecular patterns[J].Nature Reviews Immunology,2010,10(11):787-796.
- [58] BACHMANN M F,KÜNDIG T M,KALBERER C P,et al.Formalin inactivation of vesicular stomatitis virus impairs T-cell- but not T-help-independent B-cell responses[J].Journal of Virology,1993,67(7):3917-3922.
- [59] BRACIALE T J,MORRISON L A,SWEETSER M T,et al.Antigen presentation pathways to class Ⅰ and class Ⅱ MHC-restricted T lymphocytes[J].Immunological Reviews,1987,98(1):95-114.
- [60] BACHMANN M F,LUTZ M B,LAYTON G T,et al.Dendritic cells process exogenous viral proteins and virus-like particles for class Ⅰ presentation to CD8+ cytotoxic T lymphocytes[J].European Journal of Immunology,1996,26(11):2595-2600.
- [61] RUEDL C,STORNI T,LECHNER F,et al.Cross-presentation of virus-like particles by skin-derived CD8- dendritic cells:a dispensable role for TAP[J].European Journal of Immunology,2015,32(3):818-825.
- [62] AO Z J,CHEN W,TAN J,et al.Lentivirus-based virus-like particles mediate delivery of caspase 8 into breast cancer cells and inhibit tumor growth[J].Cancer Biotherapy & Radiopharmaceuticals,2019,34(1):33-41.
- [63] CHUNG Y H,CAI H,STEINMETZ N F.Viral nano-particles for drug delivery,imaging,immunotherapy,and theranostic applications[J].Advanced Drug Delivery Reviews,2020,156:214-235.
- [64] LIN M C,SHEN C H,CHANG D,et al.Inhibition of human lung adenocarcinoma growth and metastasis by JC polyomavirus-like particles packaged with an SP-B promoter-driven CD59-specific shRNA[J].Clinical Science,2019,133(21):2159-2169.
- [65] YANG J,ZHANG Q,LIU Y X,et al.Nanoparticle-based co-delivery of siRNA and paclitaxel for dual-targeting of glioblastoma[J].Nanomedicine,2020,15(14):1391-1409.
- [66] SHAN W J,ZHANG D L,WU Y L,et al.Modularized peptides modified HBc virus-like particles for encapsulation and tumor-targeted delivery of doxorubicin[J].Nanomedicine:Nanotechnology,Biology and Medicine,2018,14(3):725-734.
- [67] WOJTA-STREMAYR D,PICKL W F.Fluorosomes:fluorescent virus-like nanoparticles that represent a convenient tool to visualize receptor-ligand interactions[J].Sensors,2013,13(7):8722-8749.
- [68] LEWIS J D,DESTITO G,ZIJLSTRA A,et al.Viral nanoparticles as tools for intravital vascular imaging[J].Nature Medicine,2006,12(3):354-360.
- [69] YAN D,TENG Z D,SUN S Q,et al.Foot-and-mouth disease virus-like particles as integrin-based drug delivery system achieve targeting anti-tumor efficacy[J].Nanomedicine:Nanotechnology,Biology and Medicine,2017,13(3):1061-1070.
- [70] KINES R C,VARSAVSKY I,CHOUDHARY S,et al.An infrared dye-conjugated virus-like particle for the treatment of primary uveal melanoma[J].Molecular Cancer Therapeutics,2018,17(2):565-574.
- [71] JI T J,ZHAO Y,WANG J,et al.Tumor fibroblast specific activation of a hybrid ferritin nanocage-based optical probe for tumor microenvironment imaging[J].Small,2013,9(14):2427-2431.
- [72] SHAN W J,CHEN R H,ZHANG Q,et al.Improved stable indocyanine green(ICG)-mediated cancer optotheranostics with naturalized hepatitis B core particles[J].Adv Mater.2018,30(28):e1707567.
- [73] MASARAPU H,PATEL B K,CHARIOU P L,et al.Physalis mottle virus-like particles as nanocarriers for imaging reagents and drugs[J].Biomacromolecules,2017,18(12):4141-4153.
- [74] SHUKLA S,STEINMETZ N F.Virus-based nanomaterials as positron emission tomography and magnetic resonance contrast agents:from technology development to translational medicine[J].WIREs Nanomedicine and Nanobiotechnology,2015,7(5):708-721.
- [75] SHUKLA S,ABLACK A L,WEN A M,et al.Increased tumor homing and tissue penetration of the filamentous plant viral nanoparticle potato virus X[J].Molecular Pharmaceutics,2013,10(1):33-42.
- [76] CHACKERIAN B,RANGEL M,HUNTER Z,et al.Virus and virus-like particle-based immunogens for Alzheimer's disease induce antibody responses against amyloid-β without concomitant T cell responses[J].Vaccine,2006,24(37/38/39):6321-6331.
- [77] KUSHNIR N,STREATFIELD S J,YUSIBOV V.Virus-like particles as a highly efficient vaccine platform:diversity of targets and production systems and advances in clinical development[J].Vaccine,2012,31(1):58-83.
- [78] MIERMONT A,BARNHILL H,STRABLE E,et al.Cowpea mosaic virus capsid:a promising carrier for the development of carbohydrate based antitumor vaccines[J].Chemistry:A European Journal,2008,14(16):4939-4947.
- [79] ZHENG D W,GAO F,CHENG Q,et al.A vaccine-based nanosystem for initiating innate immunity and improving tumor immunotherapy[J].Nature Communi-cations,2020,11(1):1985.
- [80] HOOPES P J,WAGNER R J,DUVAL K,et al.Treatment of canine oral melanoma with nanotechnology-based immunotherapy and radiation[J].Molecular Pharmaceu-tics,2018,15(9):3717-3722.
- [81] KURG R,REINSALU O,JAGUR S,et al.Biochemical and proteomic characterization of retrovirus Gag based microparticles carrying melanoma antigens[J].Scientific Reports,2016,6:29425.
- [82] SIMONS B W,CANNELLA F,ROWLEY D T,et al.Bovine papillomavirus prostate cancer antigen virus-like particle vaccines are efficacious in advanced cancers in the TRAMP mouse spontaneous prostate cancer model[J].Cancer Immunology,Immunotherapy,2020,69(4):641-651.
- [83] KHOSHNEJAD M,PARHIZ H,SHUVAEV V V,et al.Ferritin-based drug delivery systems:hybrid nanocarriers for vascular immunotargeting[J].Journal of Controlled Release,2018,282:13-24.
- [84] WANG W J,LIU Z D,ZHOU X X,et al.Ferritin nanoparticle-based SpyTag/SpyCatcher-enabled click vaccine for tumor immunotherapy[J].Nanomedicine,2019,16:69-78.
- [85] SHAN W J,ZHENG H P,FU G F,et al.Bioengineered nanocage from HBc protein for combination cancer immunotherapy[J].Nano Letters,2019,19(3):1719-1727.
- [86] JEONG H,SEONG B L.Exploiting virus-like particles as innovative vaccines against emerging viral infections[J].Journal of Microbiology,2017,55(3):220-230.