(厦门大学材料学院,福建 厦门 361005)
(College of Materials,Xiamen University,Xiamen 361005,China)
BaTiO3/RGO nanocomposite; solvothermal method; microwave absorption properties
DOI: 10.6043/j.issn.0438-0479.201705029
备注
采用溶剂热法制备出BaTiO3纳米颗粒,将不同质量的BaTiO3纳米颗粒与氧化石墨烯(GO)进行复合,并在氩气保护下经过煅烧得到BaTiO3/还原氧化石墨烯(BaTiO3/RGO)纳米复合物.利用X射线衍射(XRD)、扫描电子显微镜(SEM)和透射电子显微镜(TEM)等手段系统地表征了样品的物相结构以及表面形貌,并用矢量网络分析仪(VNA)测试样品的微波吸收特性.当制备的BaTiO3/RGO纳米复合物中BaTiO3的质量分数为80.9%时,纳米复合物展现了良好的微波吸收性能; 当其厚度为2.0 mm时,在频率为10.48 GHz处的反射损耗达到-26.06 dB,且在9.32~11.54 GHz频段内反射损耗小于-10 dB.实验结果表明,BaTiO3/RGO纳米复合物具有优异的电磁波吸收性能.
Barium titanate(BaTiO3)nanoparticles were synthesized by solvothermal method and then the barium titanate/reduced graphene oxides(BaTiO3/RGO)nanocomposites with different mass ratios were obtained after calcining in argon.The structures and morphologies of samples were systematically characterized using X-ray powder diffraction(XRD),scan electron microscope(SEM)and transmission electron microscope(TEM).The microwave absorption properties of samples were tested using vector network analyzer(VNA).When the mass fraction of BaTiO3 was 80.9%,the as-prepared BaTiO3/RGO nanocomposite showed excellent microwave absorption properties,its minimum reflection loss value was -26.06 dB at 10.48 GHz with the thickness of 2.0 mm,and the bandwidth of reflection loss value less than -10 dB was from 9.32 GHz to 11.54 GHz.The results showed that BaTiO3/RGO nanocomposite had outstanding microwave absorption properties.
引言
近年来,随着无线通讯技术的发展以及雷达等微波系统的广泛应用,电磁辐射和污染日益严重[1].国内外科研工作者研究了大量不同种类的吸波材料,如碳材料[2-4]、铁氧体[5-6]以及导电高分子[7-8]等.如今,纳米技术的进步和发展为吸波材料的研究提供了新的思路.纳米介电材料可通过介电极化弛豫效应来衰减电磁波的能量,在高频波段对电磁波的吸收效果尤为显著[9-10].纳米材料具有巨大的比表面积,当处于交变电磁场中时,材料表面将产生多种极化损耗衰减电磁波能量[11-12].钛酸钡(BaTiO3)纳米颗粒作为一种典型的纳米介电材料,具有良好的化学稳定性以及高介电常数和介电响应[13-14],是一种很有发展前景的电磁波吸收材料.Zhu等[15]利用液相法制备了BaTiO3纳米管,当厚度为2.0 mm时其最小反射损耗达到了-21.8 dB; Tian等[16]将BaTiO3纳米颗粒在氢气气氛中700 ℃下煅烧4 h后,其最小反射损耗达到了-36.9 dB.但由于在高频时BaTiO3吸波材料的反射系数较大,导致其微波吸收性能不够理想[17].
还原氧化石墨烯(RGO)具有低密度、高比表面积、高电子迁移速率和优异的导热性等优点[18-19],且其特殊的二维片状结构也有利于电磁波的衰减[20-21],可作为轻重量级的吸波材料[22-23].Wen等[24]研究发现RGO的微波衰减性能优于纳米石墨片(GNs).但RGO作为吸波材料使用时,其过高的介电常数会导致反射系数较大而吸收系数较小,不能满足电磁波吸收的阻抗匹配特性[25-26].因此,国内外研究者通过将RGO与不同材料复合来调控介电常数,从而改善其阻抗匹配并提高电磁波吸收性能.Singh等[27]制备了RGO/丁腈橡胶复合物,其最小反射损耗达到-57 dB.Zong等[28]制备了Fe3O4/RGO纳米复合物,其最小反射损耗达到-44.6 dB.Wen等[29]制备了RGO/SiO2纳米复合物,在温度为473 K时其最小反射损耗达到-38 dB.因此,将纳米介电材料BaTiO3和RGO进行复合制备的BaTiO3/RGO纳米复合物,不仅可以通过改变其复介电常数来改善材料的阻抗匹配特性,而且随着RGO的引入,BaTiO3/RGO纳米复合物将形成更多的表界面,由于界面极化效应以及协同效应的作用,其微波吸收性能将得到有效加强,比单一组分的吸波材料具有明显的优势.
本研究采用溶剂热法制备BaTiO3纳米颗粒,将不同质量BaTiO3纳米颗粒与氧化石墨烯(GO)进行复合,并在氩气保护下经过煅烧得到一系列BaTiO3/RGO纳米复合物,并对其物相结构和表面形貌进行表征,测试其微波吸收特性.
1 实验部分
1.1 试 剂乙醇、四氯化钛(TiCl4)、八水合氢氧化钡(Ba(OH)2·8H2O)、氢氧化钠(NaOH)、十六烷基三甲基溴化铵(CTAB)以及乙二胺均为分析纯,购自国药集团化学试剂有限公司.采用Hummers法[25]合成GO.去离子水为实验室自制.
1.2 BaTiO3/RGO纳米复合物的制备取220 μL TiCl4分散于20 mL乙醇中,配制成TiCl4的乙醇溶液; 取1.241 6 g Ba(OH)2·8H2O溶于20 mL去离子水中,超声30 min使其完全溶解; 然后将TiCl4的乙醇溶液逐滴加入Ba(OH)2溶液中,并滴加0.2 mol/L的NaOH溶液,使混合溶液pH>13; 搅拌120 min后将溶液转入100 mL的聚四氟乙烯内衬的反应釜中,并将反应釜置于真空干燥箱中240 ℃下保温24 h进行溶剂热反应.反应结束后,离心收集产物并用去离子水和无水乙醇交替洗涤3次,冷冻干燥24 h后得到BaTiO3纳米颗粒.
将40.0 mg GO在超声条件下分散于20 mL 去离子水中,形成均匀的棕褐色溶液; 然后加入160 mg BaTiO3纳米颗粒和6.0 mg CTAB,超声2 h使其形成均匀的悬浮液; 往悬浮液中滴加5 mL乙二胺,搅拌12 h后转入50 mL的聚四氟乙烯内衬的反应釜中,并将反应釜置于真空干燥箱中200 ℃下保温180 min进行溶剂热反应.反应结束后,离心收集产物并用去离子水和无水乙醇交替洗涤3次,冷冻干燥24 h后得到样品.随后将样品在氩气气氛下600 ℃退火处理120 min得到最终产物,记为S1.其他实验条件不变,只改变加入的BaTiO3质量为93和60 mg,制得的产物分别记为S2和S3; 不加入GO时得到的产物记为S0.
1.3 样品测试采用德国Bruker公司D8 Advance X射线衍射(XRD)仪分析样品的物相结构,Cu Kα的波长为0.154 18 nm,工作电压为40 kV,工作电流为40 mA,测试的角度范围为5°~90°,测量模式为平板扫描模式.使用日本日立仪器公司SU70场发射扫描电子显微镜(SEM)和日本电子株式会所JEM-1400透射电子显微镜(TEM)表征样品表面形貌特征.
采用美国TA仪器公司SDT Q600热重/差热联用热分析仪,在空气气氛下对样品进行热重分析,加热速率为10 ℃/min.
采用同轴线法测量样品的微波吸收特性,测量时先将样品与石蜡混合(样品的质量分数为40%),压制成外径为7.00 mm,内径为3.04 mm的圆环; 然后使用Agilent Technologies公司N5222A型矢量网络分析仪(vector network analyzer,VNA)测试样品的电磁参数,其测试频率范围为2~18 GHz,并根据电磁参数计算出样品的反射损耗.
2 结果及讨论
2.1 BaTiO3/RGO纳米复合物的结构与形貌图1(a)和(b)分别为BaTiO3纳米颗粒的TEM和SEM图,从图中可以看出,制备的BaTiO3呈规则的球形,其尺寸约为70 nm,粒径较为均一,且分散性良好,并未发现团聚现象.图1(c)和(d)分别为BaTiO3/RGO纳米复合物的TEM和SEM图,从图中可以看出,BaTiO3纳米颗粒均匀负载在RGO纳米片上且其尺寸保持不变.
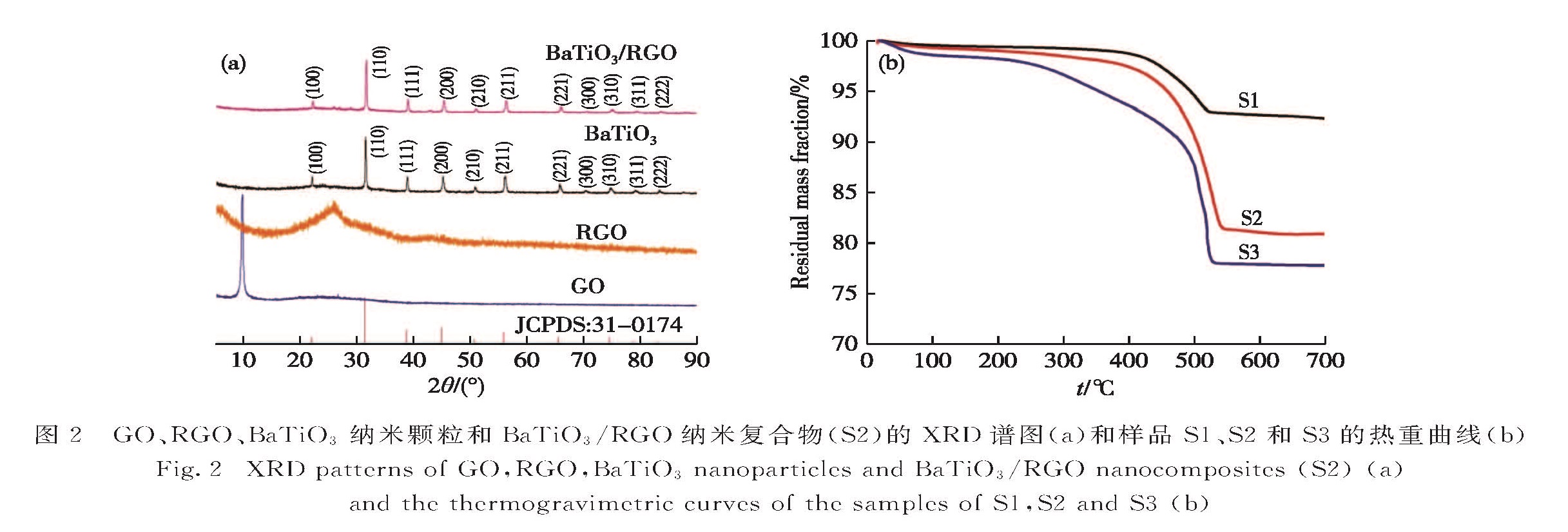
图2(a)分别为GO、RGO、BaTiO3纳米颗粒和BaTiO3/RGO纳米复合物(S2)的XRD谱图.GO在2θ=9.8°处有一个强烈衍射峰,表明GO具有有序的层状结构[26-27].RGO在2θ=26.0°存在一个较弱的衍射峰,这与文献[30]中RGO特征峰的位置相符.BaTiO3纳米颗粒的衍射峰都较为尖锐,且在2θ=21.99°,31.56°,38.87°,45.20°,50.89°,56.09°,65.77°,70.30°,74.92°,79.06°以及83.30°的衍射峰对应于立方相BaTiO3(JCPDS:31-0174)的(100),(110),(111),(200),(210),(211),(221),(300),(310),(311)和(222)晶面,这表明溶剂热法制备的BaTiO3纳米颗粒的结晶性良好且为立方相结构.在BaTiO3/RGO纳米复合物的XRD谱图中只观察到BaTiO3的衍射峰,这是由于BaTiO3纳米颗粒的衍射峰很强而RGO衍射峰强度较低[31]; 而BaTiO3衍射峰的峰位没有改变,说明复合后BaTiO3纳米颗粒的晶体结构并没有发生变化.由图2(b)可知,温度高于
图1 BaTiO3纳米颗粒(a、b)和BaTiO3/RGO纳米复合物(c、d)的TEM(a、c)和SEM(b、d)图
Fig.1 TEM(a,c)and SEM(b,d)images of BaTiO3 nanoparticles(a,b)and BaTiO3/RGO nanocomposites(c,d)图2 GO、RGO、BaTiO3纳米颗粒和BaTiO3/RGO纳米复合物(S2)的XRD谱图(a)和样品S1、S2和S3的热重曲线(b)
Fig.2 XRD patterns of GO,RGO,BaTiO3 nanoparticles and BaTiO3/RGO nanocomposites(S2)(a)and the thermogravimetric curves of the samples of S1,S2 and S3(b)550 ℃后样品的热重曲线趋于水平,说明S1、S2和S3中BaTiO3的质量分数分别为92.3%,80.9%和77.8%,对应的RGO的质量分数分别为7.7%,19.1%和22.2%.
2.2 BaTiO3/RGO纳米复合物的吸波特性图3(a)和(b)显示了不同样品的复介电常数实部ε'和虚部ε″在频率2~18 GHz内的变化规律.从图中可以看出S0、S1、S2和S3的ε'和ε″的值依次增大,表明ε'和ε″随着样品中RGO质量分数的升高而增大,这是由于与BaTiO3纳米颗粒相比,RGO的介电常数较大[22].由图3(a)可知,随着频率升高,样品的ε'值整体上呈减小趋势.其中,S0的ε'值变化幅度较小,在3.70附近波动; S1的ε'值从7.20减至6.41; S2的ε'值从18.67减至12.51; 而S3的ε'值则从23.98减至12.91.由此可以看出,S0和S1的弛豫现象并不明显,而随着RGO质量分数的增加,S2和S3产生了明显的弛豫现象.由图3(b)可知,随着频率升高,S1、S2和S3的ε″值整体上呈先减小后增大的趋势.上述结果表明,可通过调整样品中BaTiO3和RGO的质量分数,来调控BaTiO3/RGO纳米复合物的复介电常数,进而增加材料的极化特性,改善其阻抗匹配.
图3 样品S0、S1、S2和S3的ε'(a)、ε″(b)、tan δε(c)和Cole-Cole半圆(d)
Fig.3 The ε'(a),ε″(b),tan δε(c)and the Cole-Cole semicircles(d)of the samples of S0,S1,S2 and S3吸波材料的介电损耗角正切值tan δε=ε″/ε',可用来表征其介电损耗的大小[32].图3(c)为不同样品的损耗角正切值在频率2~18 GHz内的变化规律.当频率升高时,S1、S2和S3的损耗角正切值呈现先减小后增大的趋势.当频率为2 GHz时,S1、S2和S3的损耗角正切值依次为0.23,0.59和0.74.这表明随着GO质量分数的增大,制得的BaTiO3/RGO纳米复合物的损耗角正切值逐渐增大.这是由于GO与BaTiO3复合时,BaTiO3/RGO纳米复合物在电磁波中将发生界面极化[33],RGO与其负载的BaTiO3纳米颗粒间产生电荷转移后引入载流子的运动,引起介电损耗.当GO质量分数增大时,制备的BaTiO3/RGO纳米复合物形成更丰富的表界面,从而界面极化产生更强的介电损耗.图3(d)展示了样品S0、S1、S2和S3的复介电常数虚部ε″随实部ε'的变化关系.一般通过Cole-Cole半圆来判断材料是否产生德拜弛豫[34].从图中可以看出,样品S2和S3存在明显的弛豫现象.
为了进一步衡量样品的吸波性能,先计算出微波吸收层的归一化输入阻抗Zin,其计算式为
Zin=((μr)/(εr))1/2tanh[i(2πft)/c(μrεr)1/2],
再由下式计算出样品的反射损耗RL[35-36]:
RL=20lg|(Zin-1)/(Zin+1)|.
其中,c为微波在真空中的传播速度,μr和εr分别为吸波材料的复磁导率和复介电常数,t为微波吸收层的厚度,i为复介电常数表达式εr=ε'-iε″中的虚数单位.考虑到BaTiO3/RGO纳米复合物的磁性极为微弱,样品的复磁导率μr可近似为1.当反射损耗低于-10 dB时,可认为约10%的微波被反射,剩余的90%被吸收.
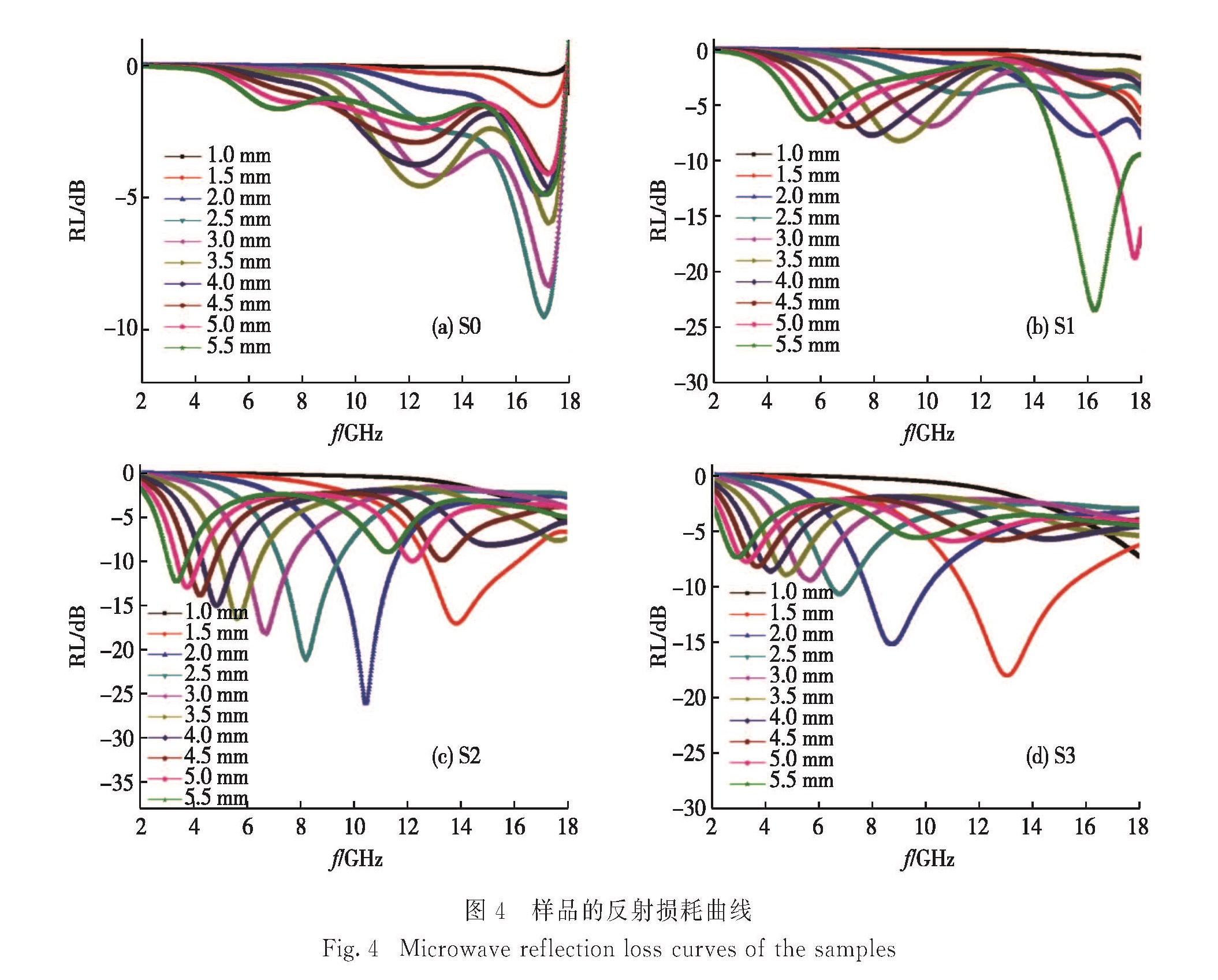
图4(a)~(d)分别为S0、S1、S2和S3在2~18 GHz频率范围内,不同厚度样品的理论反射损耗.样品S0的最小反射损耗仅为-9.47 dB,S1、S2和S3的微波吸收性能相较于S0有明显的提高.其中,当厚度为5.5 mm时,S1的最小反射损耗为-23.52 dB,且在14.96~17.68 GHz频段内反射损耗小于-10 dB; 当厚度为2.0 mm时,S2的最小反射损耗为-26.06 dB,且在9.32~11.54 GHz频段内反射损耗小于-10 dB; 当厚度为1.5 mm时,S3的最小反射损耗为-18.16 dB,且在11.35~15.29 GHz频段内反射损耗小于-10 dB.S1与S2的最小反射损耗和反射损耗小于-10 dB对应的频段宽度较为接近,但S1的厚度大于S2,故S1的微波吸收性能不如S2.虽然S3的厚度小于S2,且S3反射损耗小于-10 dB对应的频段宽度大于S2,但S3的最小反射损耗大于S2,故S3的微波吸收性能也不如S2.综合考虑样品的厚度、反射损耗的大小和反射损耗小于-10 dB对应的频段宽度3个参数,可以发现,样品S2展现了最好的微波吸收性能.此外,由图4(c)可知,当吸波层厚度增大,S2的反射损耗峰往低频方向移动,这是由于1/4波长相消理论产生的频移现象.样品S2能表现出优异的吸波性能,主要由于:1)高介电常数的RGO可以调节介电常数的大小,从而改善阻抗匹配,使更多的电磁波进
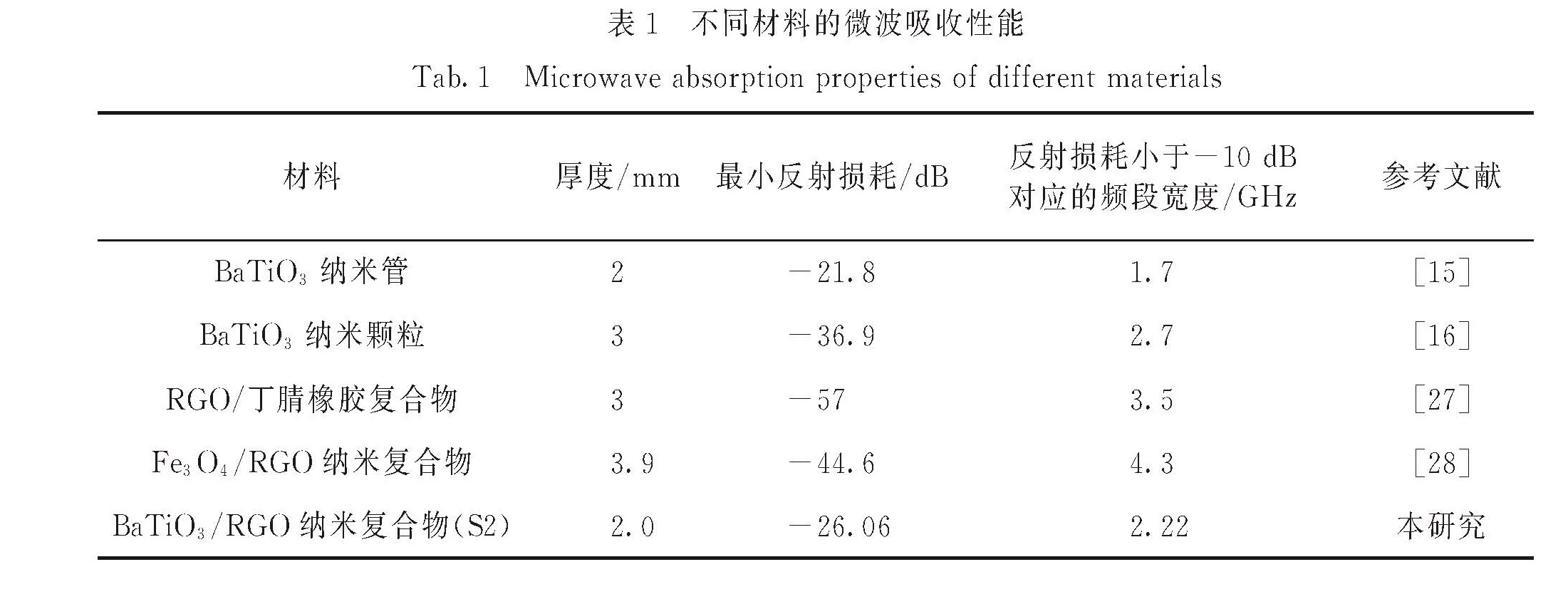
入吸波体内,拓宽吸收频带范围; 2)化学法合成的RGO表面带有官能团并存在缺陷,在电磁场下易产生极化[37-38]; 3)二维RGO与BaTiO3纳米颗粒构建的三维导电网络有利于电磁波能量的转化与衰减; 4)BaTiO3纳米颗粒与RGO产生的界面能有效地增加界面极化效应,增加电磁波能量的吸收[39].表1显示了不同材料的微波吸收性能,综合对比可知,在厚度较小的情况下,本研究制备的BaTiO3/RGO纳米复合物(S2)具有较好的吸波性能.
3 结 论
本研究中采用去离子水和乙醇作为溶剂进行溶剂热反应,成功制备出尺寸约为70 nm的BaTiO3纳米颗粒.利用CTAB和乙二胺作为表面活性剂,将不同质量的BaTiO3纳米颗粒与GO复合并在氩气气氛中煅烧120 min后得到BaTiO3/RGO纳米复合物.研究发现,通过改变加入的BaTiO3和GO的质量比,可以调控BaTiO3/RGO纳米复合物复介电常数的大小和介电损耗角正切值.当制备的BaTiO3/RGO纳米复合物中BaTiO3的质量分数为80.9%时展现出良好的微波吸收性能,当其厚度为2.0 mm时,在频率为10.48 GHz处的反射损耗达到为-26.06 dB,且在9.32~11.54 GHz频段内的反射损耗小于-10 dB.实验结果表明,BaTiO3/RGO纳米复合物是一种有应用前景的微波吸收材料.
- [1] MAOQIONG L I.Current status and future trends of electromagnetic-wave absorbent with nano-structure [J].Materials Review,2002,16(9):15-17.
- [2] LV H,GUO Y,ZHAO Y,et al.Achieving tunable electromagnetic absorber via graphene/carbon sphere composites [J].Carbon,2016,110:130-137.
- [3] CAO M S,SONG W L,HOU Z L,et al.The effects of temperature and frequency on the dielectric properties,electromagnetic interference shielding and microwave-absorption of short carbon fiber/silica composites [J].Carbon,2010,48(3):788-796.
- [4] CAO M S,YANG J,SONG W L,et al.Ferroferric oxide/multiwalled carbon nanotube vs polyaniline/ferroferric oxide/multiwalled carbon nanotube multiheterostructures for highly effective microwave absorption [J].ACS Applied Materials & Interfaces,2012,4(12):6949-6956.
- [5] KHAN M A,ULLAH H,JUNAID M,et al.Structural,magnetic and dielectric properties of Yb3+ doped BaCo-X hexagonal nanoferrites [J].Journal of Alloys and Compounds,2017,695:3674-3681.
- [6] SINGH L,MALHOTRA J,SINGH C,et al.Investigation of microwave and electrical characteristics of Co-Zr substituted M-type Ba-Sr hexagonal ferrite [J].Materials Science-Poland,2015,33(2):335-339.
- [7] THOMASSIN J M,JÉRÔME C,PARDOEN T,et al.Polymer/carbon based composites as electromagnetic interference(EMI)shielding materials [J].Materials Science and Engineering R,2013,74(7):211-232.
- [8] CHEN Y,ZHANG H B,HUANG Y Q,et al.Magnetic and electrically conductive epoxy/graphene/carbonyl iron nanocomposites for efficient electromagnetic interference shielding [J] Composites Science and Technology,2015,118:178-185.
- [9] JONSCHER A K.Dielectric relaxation in solids [J].Journal of Physics D:Applied Physics,1999,32(14):R57-R70.
- [10] CAO M S,SHI X L,FANG X Y,et al.Microwave absorption properties and mechanism of cagelike ZnO/SiO2 nanocomposites [J].Applied Physics Letters,2007,91(20):203110.
- [11] WANG Z J,WU L N,ZHOU J G,et al.Magnetite nanocrystals on multiwalled carbon nanotubes as a synergistic microwave absorber [J].The Journal of Physical Chemistry C,2013,117(10):5446-5452.
- [12] LIANG Y,LI Y,WANG H,et al.Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction [J].Nature Materials,2011,10(10):780-786.
- [13] YUN W S,URBAN J J,GU Q,et al.Ferroelectric pro-perties of individual barium titanate nanowires investigated by scanned probe microscopy [J].Nano Letters,2002,2(5):447-450.
- [14] SAKABE Y,YAMASHITA Y,YAMAMOTO H.Di-electric properties of nano-crystalline BaTiO3 synthesized by micro-emulsion method [J].Journal of the European Ceramic Society,2005,25(12):2739-2742.
- [15] ZHU Y F,ZHANG L,NATSUKI T,et al.Facile synthesis of BaTiO3 nanotubes and their microwave absorption properties [J].ACS Applied Materials & Interfaces,2012,4(4):2101-2106.
- [16] TIAN L H,YAN X D,XU J L,et al.Effect of hydroge-nation on the microwave absorption properties of BaTiO3 nanoparticles [J].Journal of Materials Chemistry A,2015,3(23):12550-12556.
- [17] PETROV V M,GAGULIN V V.Microwave absorbing materials [J].Inorganic Materials,2001,37(2):93-98.
- [18] AVOURIS P,CHEN Z,PEREBEINOS V.Carbon-based electronics [J].Nature Nanotechnology,2007,2(10):605-615.
- [19] BALANDIN A A.Thermal properties of graphene and nanostructured carbon materials [J].Nature Materials,2011,10(8):569-581.
- [20] 贾宝富,刘述章,林为干.反雷达涂覆材料中吸收粒子的最佳外形[J].电子科技大学学报,1990,19(4):342-345.
- [21] YOSHIDA S,ANDO S,SHIMADA Y,et al.Crystal structure and microwave permeability of very thin Fe-Si-Al flakes produced by microforging [J].Journal of Applied Physics,2003,93(10):6659-6661.
- [22] LIAO L,PENG H,LIU Z.Chemistry makes graphene beyond graphene [J].Journal of the American Chemical Society,2014,136(35):12194-12200.
- [23] LI Q,MAHMOOD N,ZHU J,et al.Graphene and its composites with nanoparticles for electrochemical energy applications [J].Nano Today,2014,9(5):668-683.
- [24] WEN B,WANG X X,CAO W Q,et al.Reduced graphene oxides:the thinnest and most lightweight materials with highly efficient microwave attenuation performances of the carbon world [J].Nanoscale,2014,6(11):5754-5761.
- [25] WANG L,HUANG Y,SUN X,et al.Synthesis and microwave absorption enhancement of graphene@Fe3O4@SiO2@NiO nanosheet hierarchical structures [J].Nanoscale,2014,6(6):3157-3164.
- [26] SUN X,HE J,LI G,et al.Laminated magnetic graphene with enhanced electromagnetic wave absorption properties [J].Journal of Materials Chemistry C,2013,1(4):765-777.
- [27] SINGH V K,SHUKLA A,PATRA M K,et al.Microwave absorbing properties of a thermally reduced graphene oxide/nitrile butadiene rubber composite [J].Carbon,2012,50(6):2202-2208.
- [28] ZONG M,HUANG Y,ZHAO Y,et al.Facile preparation,high microwave absorption and microwave absorbing mechanism of RGO-Fe3O4 composites [J].RSC Advances,2013,3(45):23638-23648.
- [29] WEN B,CAO M,LU M,et al.Reduced graphene oxides:light-weight and high-efficiency electromagnetic interference shielding at elevated temperatures [J].Advanced Materials,2014,26(21):3484-3489.
- [30] HUMMERS W S,JR,OFFEMAN R E.Preparation of graphitic oxide [J].Journal of the American Chemical Society,1958,80(6):1339.
- [31] XIAN T,YANG H,DI L,et al.Photocatalytic reduction synthesis of SrTiO3-graphene nanocomposites and their enhanced photocatalytic activity [J].Nanoscale Research Letters,2014,9(1):327.
- [32] XU P,HAN X,WANG C,et al.Synthesis of electromagnetic functionalized nickel/polypyrrole core/shell composites [J].The Journal of Physical Chemistry B,2008,112(34):10443-10448.
- [33] 赵东林,沈曾民.含碳纳米管微波吸收材料的制备及其微波吸收性能研究[J].无机材料学报,2005,20(3):608-612.
- [34] COLE K S,COLE R H.Dispersion and absorption in dielectrics Ⅰ.Alternating current characteristics [J].Journal of Chemical Physics,1941,9(4):341-351.
- [35] SINGH P,BABBAR V K,RAZDAN A,et al.Complex permittivity,permeability,and X-band microwave absorption of CaCoTi ferrite composites [J].Journal of Applied Physics,2000,87(9):4362-4366.
- [36] MILES P A,WESTPHAL W B,VON HIPPEL A.Di-electric spectroscopy of ferromagnetic semiconductors [J].Reviews of Modern Physics,1957,29(3):279-307.
- [37] CAO W Q,WANG X X,YUAN J,et al.Temperature dependent microwave absorption of ultrathin graphene composites [J].Journal of Materials Chemistry C,2015,3:10017-10022.
- [38] DOU Y K,LI J B,FANG X Y,et al.The enhanced polarization relaxation and excellent high-temperature di-electric properties of N-doped SiC [J].Applied Physics Letters,2014,104(5):052102.
- [39] YANG H J,CAO W Q,ZHANG D Q,et al.NiO hierarchical nanorings on SiC:enhancing relaxation to tune microwave absorption at elevated temperature [J].ACS Applied Materials & Interfaces,2015,7(13):7073-7077.